Solid StateHard
Question
MgAl2O4, is found in the Spinal structure in which O2- ions constitute CCP lattice, Mg2+ ions occupy 1/8th of the Tetrahedral voids and Al3+ ions occupy 1/2 of the Octahedral voids.
Find the total +ve charge contained in one unit cell.
Find the total +ve charge contained in one unit cell.
Options
A.+7/4 electronic charge
B.+6 electronic charge
C.+2 electronic charge
D.+8 electronic charge
Solution
Total positive charge = Charge on Mg2+ + Charge on Al3+
=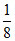 × 8 × 2 +
× 8 × 2 + 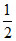 × 4 × 3 = 8 electronic charge.
× 4 × 3 = 8 electronic charge.
=
Create a free account to view solution
View Solution FreeMore Solid State Questions
If the positions of Na+ and Cl- are interchanged in NaCl, the crystal lattice with respect to Na+ and CI- is...For a certain crystal, the unit cell axial lengths are found to be a = 5.62 Å, b = 7.41 Å and c = 10.13 Å. The three coo...A transition metal M can exist in two oxidation states +2 and +3. It forms an oxide whose experimental formula is given ...A molecule A2B (Mw = 166.4) occupies triclinic lattice with a = 5, b = 8, and c = 4 .If the density of AB2 is 5.2 g cm-3...MgO exist in a rock-salt type unit cell. Each Mg+2 ion will be in contact with...