Solid StateHard
Question
A molecule A2B (Mw = 166.4) occupies triclinic lattice with a = 5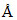 , b = 8
, b = 8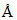 , and c = 4
, and c = 4 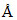 .
.
If the density of AB2 is 5.2 g cm-3, the number of molecules present in one unit cell is
If the density of AB2 is 5.2 g cm-3, the number of molecules present in one unit cell is
Options
A.2
B.3
C.4
D.5
Solution
Volume of unit cell = a × b × c = 5 × 10-8 × 8 × 10-8 × 4 × 10-8 = 1.6 × 10-22 cm3
Mass of unit cell = 1.6 × 10-22 × 5.2 = 8.32 × 10-22 g
Number of molecules in one unit cell =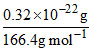 = 3
= 3
Mass of unit cell = 1.6 × 10-22 × 5.2 = 8.32 × 10-22 g
Number of molecules in one unit cell =
Create a free account to view solution
View Solution FreeMore Solid State Questions
The total number of 2nd nearest Cl- ions to the Na+ ion in NaCl unit cell are -...The spinal structure (AB2O4) consists of an fcc array of O2- ions in which the :...A metal having atomic mass 60.22 g/mole crystallizes in ABCABC…. type packing. The density of each metal atom if the edg...The radius ratio of P2+ and Q- ions is 0.8. The unit cell of this ionic solid is...Distance between two tetrahedral voids in FCC unit cell is -...