Solid StateHard
Question
How many units cells are there in 1.00 g cube shaped ideal crystal of AB (Mw = 60) which has a NaCl type lattice
Options
A.6.02 × 1023
B.1.00 × 2022
C.2.50 × 1021
D.6.02 × 1024
Solution
Z = 4, M = 60
60 × 4 gram _________________ 6.023 × 1023 unit cells
1 gram _________________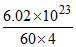 = 2.5 × 1021
= 2.5 × 1021
60 × 4 gram _________________ 6.023 × 1023 unit cells
1 gram _________________
Create a free account to view solution
View Solution FreeMore Solid State Questions
The crystalline structure of a solid is CCP for ‘X’ atoms. If ‘Y’ atoms occupy all the octahedral voids and ‘Z’ atoms oc...Which of the following statement(s) is/are incorrect regarding the defects in solids?...Lead metal has a density of 11.34 g/cm3 and crystallizes in a face-centered lattice. Choose the correct alternatives...How many unit cell are present in a cube-shaped ideal crystal of NaCl of mass 1.00 gm. [Atomic masses : Na = 23, Cl = 35...A binary solid $(A^+ B^-)$ has a rock salt structure. If the edge length is $400 \text{ pm}$ and the radius of the catio...