Solid StateHard
Question
Lead metal has a density of 11.34 g/cm3 and crystallizes in a face-centered lattice. Choose the correct alternatives
Options
A.the volume of one unit cell is 1.214 × 10-22 cm3
B.the volume of one unit cell is 1.214 × 10-19 cm3
C.the atomic radius of lead is 175 pm
D.the atomic radius of lead is 155.1 pm
Solution
Density = 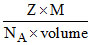 so, Volume =
so, Volume = 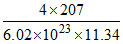 = 1.213 × 10-22 cm3; 4r = a√2
= 1.213 × 10-22 cm3; 4r = a√2
r =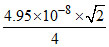 = 175 pm
= 175 pm
Volume = a3 = 1.213 × 10-22
so, a = (1.213 × 10-24)1/3
a = 4.95 × 10-8 cm.
r =
Volume = a3 = 1.213 × 10-22
so, a = (1.213 × 10-24)1/3
a = 4.95 × 10-8 cm.
Create a free account to view solution
View Solution FreeMore Solid State Questions
Which of the following may have Frenkel defect?...The number of octahedral void(s) per atom present in a cubic close-packed structure is...Which of the following statement(s) is/ are correct with respect to zinc blende structure?...MgO exists in a rock salt type unit cell. Each $\text{Mg}^{2+}$ ion will be in contact with:...In a face centerd lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers. Then the for...