Solid StateHard
Question
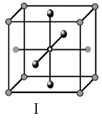
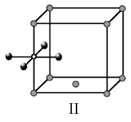
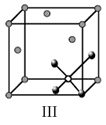
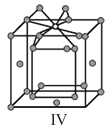
Options
A.I and II represent tetrahedral holes
B.II, III and IV represent tetrahedral holes
C.I and II represent octahedral holes
D.I, II and IV represent octahedral holes
More Solid State Questions
The density of KBr is 2.75 g-cm-3 and length of the unit cell is 654 pm (K = 39 and Br = 80) then what is true about the...Glass is a...CsCl crystallises in body centred cubic lattice. If ′a′ is its edge length then which of the following expre...Which of the following is a ferromagnetic substance?...Select the correct co-ordinates for octahedral void in FCC unit cell...