Solid StateHard
Question
Figure shows a cube of unit cell of CCP arrangement with face centered atoms marked 1, 2, 3.Which of the following statements is true.
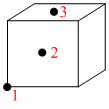

Options
A.Atom 3 is twice as far from 1 as from 2
B.Atom 2 is equidistant from atoms 1 & 3.
C.Atom 2 is nearer to 1 than to 3.
D.All atoms lie on a right angled triangle.
More Solid State Questions
NaCl crystal has which type of crystal structure?...If the anions (A) form hexagonal closest packing and cations (C) occupy only 2/3 octahedral voids in it, then the genera...Sodium oxide has anti-fluorite structure. The percentage of the tetrahedral voids occupied by the sodium ions is...In a CCP lattice of X and Y, X atoms are present at the corners while Y atoms are at face centers.Then the formula of th...Superconductors are substances which...