Atomic StructureHard
Question
The increasing order (lowest first) for the values of e/m (charge /mass) for electron (p),neutron (n) and alpha particle (α) is :
Options
A.e, p, n, α
B.n, p, e, α
C.n, p, α, e
D.n, α, p, e
Solution
Particle = n α p e
e/m value =
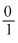
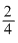
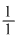
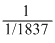
mass is taken in amu.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following ion have the same number of unpaired electrons as in Fe2+ (Z = 26)?...The correct order of atomic radii in group 13 elements is...Atoms have void spaces. It was first suggested by...Azimuthal quantum number defines...For a photochemical reaction A → B, 1 × 10–5 moles of ‘B’ were formed on absorption of 6.626 × 107 erg at 360 nm. The qu...