Atomic StructureHard
Question
Solubility product of silver bromide is 5.0 × 10-13. The quantity of potassium bromide (molar mass taken as 120 g of mol-1) to be added to 1 litre of 0.05 M solution of silver nitrate to start the precipitation of AgBr is
Options
A.1.2 × 10-10 g
B.1.2 × 10-9 g
C.6.2 × 10-5 g
D.5.0 × 10-8 g
Solution
Ag+ + Br- ⇋ AgBr
Precipitation starts when ionic product just exceeds solubility product
Ksp = [Ag+][Br-]
[Br-] =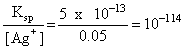
i.e., precipitation just starts when 10-11 moles of KBr is added to 1L of AgNO3 solution.
No. of moles of KBr to be added = 10-11
∴ weight of KBr to be added = 10-11 × 120
= 1.2 × 10-9g
Precipitation starts when ionic product just exceeds solubility product
Ksp = [Ag+][Br-]
[Br-] =
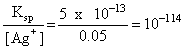
i.e., precipitation just starts when 10-11 moles of KBr is added to 1L of AgNO3 solution.
No. of moles of KBr to be added = 10-11
∴ weight of KBr to be added = 10-11 × 120
= 1.2 × 10-9g
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
If the numbers of orbitals of a particular type were (3l +1), but spin quantum numbers were only +1/2 and –1/2, then d-t...In which of these options do both constituents of the pair have the same magnetic moment?...What is the wave number of the radiation of lowest frequency in the Lyman series of the spectrum of Li2+ ion?...The eyes of a certain member of the reptile family pass a single visual single to the brain when the visual receptors ar...Which of the given statement (s) is/are false.I Orbital angular momentum of the electron having n = 5 and having value o...