Atomic StructureHard
Question
Which of the given statement (s) is/are false.
I Orbital angular momentum of the electron having n = 5 and having value of the azimuthal quantum number as lowest for this principle quantum number is h/π.
II If n = 3, l = 0, m = 0, for the last valence shell electron, then the possible atomic number must be 12 or 13.
III Total spin of electrons for the atom 25Mn is ± 7/2.
IV Spin magnetic moment of inert gas is 0
I Orbital angular momentum of the electron having n = 5 and having value of the azimuthal quantum number as lowest for this principle quantum number is h/π.
II If n = 3, l = 0, m = 0, for the last valence shell electron, then the possible atomic number must be 12 or 13.
III Total spin of electrons for the atom 25Mn is ± 7/2.
IV Spin magnetic moment of inert gas is 0
Options
A.I, II and III
B.II and III only
C.I and IV only
D.None of these
Solution
I : For n = 5, lmin = 0. ∴ Orbital angular momentum = 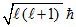 = 0.(False)
= 0.(False)
II : Outermost electronic configuration = 3s1 or 3s2. ∴ possible atomic number = 11or 12 (False).
III : Mn25 = [Ar] 3d5 4s2. ∴ 5 unpaired electrons ∴ Total spin = ±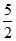 (False).
(False).
IV : Inert gases have no unpaired electrons. ∴ spin magnetic moment = 0 (True).
II : Outermost electronic configuration = 3s1 or 3s2. ∴ possible atomic number = 11or 12 (False).
III : Mn25 = [Ar] 3d5 4s2. ∴ 5 unpaired electrons ∴ Total spin = ±
IV : Inert gases have no unpaired electrons. ∴ spin magnetic moment = 0 (True).
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The de Broglie wavelength of electron of He+ ion is 3.32 Å. If the photon emitted upon de-excitation of this He+ ion is ...X X1 + H2O → HX1 + HOXSelect the correct statement for above reaction :...Given,(A) (B) n=3,1=2, m1=-1, ms=+12 The maximum number of electron(s) in an atom that can have the quantum numbers as g...Which is the correct order of ionic sizes ?(Atomic Number : Ce = 58, Sn = 50, Yb = 70 and Lu = 71)...The de Broglie wavelength associated with a ball of mass 1 kg having kinetic energy 0.5 J is :...