Ionic EquilibriumHard
Question
Three reactions involving H2PO4- are given below :
(i) H3PO4 + H2O → H3O+ + H2PO4-
(ii)H2PO4- + H2O → HPO42- + H3O+
(iii) H2PO4- + OH- → H3PO4 + O2-
In which of the above does H2PO4- act as an acid ?
(i) H3PO4 + H2O → H3O+ + H2PO4-
(ii)H2PO4- + H2O → HPO42- + H3O+
(iii) H2PO4- + OH- → H3PO4 + O2-
In which of the above does H2PO4- act as an acid ?
Options
A.(ii) only
B.(i) and (ii)
C.(iii) only
D.(i) only
Solution
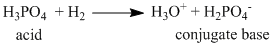

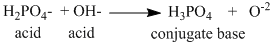
Only in reaction (ii) H2PO4- acids as ′acid′.
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Which of the following is not correct :...The solubility of AgCl in water is 0.001435 g per litre at 15o C. The solubility product of AgCl is(Ag = 108, Cl = 35.3)...The indicator constant for an acidic indicator, HIn is 5 × 10−6 M. This indicator appears only in the colour of acidic f...A volume of 20 ml of 0.8 M-HCN solution is mixed with 80 ml of 0.4 M-NaCN solution. Calculate the pH of the resulting so...The pH of 4.0 × 10−4 M-HNO3 solution is (log 2 = 0.3)...