Solid StateHard
Question
A compound of A and B crystallizes in a cubic lattice in which A atoms occupy the lattice points at the corners of a cube and two atoms of B occupy the center of each of the cube faces. What is the formula of this compound ?
Options
A.AB3
B.AB4
C.AB2
D.AB6
Solution
for A, 8 × 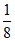 = 1
= 1
for B, 12 ×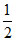 = 6
= 6
so AB6
for B, 12 ×
so AB6
Create a free account to view solution
View Solution FreeMore Solid State Questions
An ionic solid of XY type having anions in CCP lattice and cations in the octahedral voids.Let a be the edge length of a...The ionic radii of Cs and Cl are 0.165 nm and 0.181nm respectively. Their atomic weights are 133 and 35.5. Then,...Distance between two tetrahedral voids in FCC unit cell is -...In a solid lattice, the cation has left a lattice site and is located at interstitial position, the lattice defect is :...An element (atomic mass = 100 g/mole) having BCC structure has unit cell edge 400 pm. The density of the element is (no....