Atomic StructureHard
Question
n-Butane (C4H10) is produced by monobromination of C2H6 followed by Wurtz reaction. Calculate the volume of ethane at STP required to produce 55 g of n-butane. The bromination takes place with 90% yield and the Wurtz reaction with 85% yield.
Options
A.27.75 L
B.55.5 L
C.55.5 L
D.5.55 L
Solution
2C2H6 + Br2 → 2C2H5Br + HBr
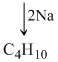
12 × 4 + 10 = 58 g
58 g of n-butane ⇒ 2 × 22.4 L of C2H6 at STP
⇒ 2 × 22.4 ×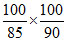
58 g of n-butane = 58.56 L
58 g of n-butane = 55.5 L
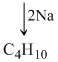
12 × 4 + 10 = 58 g
58 g of n-butane ⇒ 2 × 22.4 L of C2H6 at STP
⇒ 2 × 22.4 ×
58 g of n-butane = 58.56 L
58 g of n-butane = 55.5 L
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
The atomic nucleus contains :...Which one is a wrong statement ?...According to Bohr′s model of hydrogen atom the electric current generated due to motion of electron in nth orbit i...Let υ1 be the frequency of the series limit of the Lyman series, υ2 be the frequency of the first line of th...Given that abundances of isotopes 54Fe, 56Fe, and 57Fe are 5%, 90% and 5% respectively, the atomic mass of Fe is :-...