Atomic StructureHard
Question
In aqueous solution the ionization constants for carbonic acid are
K1 = 4.2 × 10-7 and K2 = 4.8 × 10-11
Select the correct statement for a saturated 0.034 M solution of the carbonic acid.
K1 = 4.2 × 10-7 and K2 = 4.8 × 10-11
Select the correct statement for a saturated 0.034 M solution of the carbonic acid.
Options
A.The concentration of CO32- is 0.034 M.
B.The concentration of CO32- is greater than that of HCO3-.
C.The concentration of H+ and HCO3- are approximately equal.
D.The concentration of H+ is double that of CO32-.
Solution
A → H2CO3 ⇋ H+ + HCO3- K1 = 4.2 × 10-7
B → HCO3- ⇋ H+ + CO3-2 K2 = 4.8 × 10-11
As K2 ≪ K1
All major [H+]total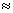 [H+]A
[H+]A
and from I equilibrium, [H+]A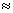 [HCO3-]
[HCO3-] 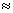 [H+]total
[H+]total
[CO3-2] is negligible compared to [HCO3-] or [H+]total
B → HCO3- ⇋ H+ + CO3-2 K2 = 4.8 × 10-11
As K2 ≪ K1
All major [H+]total
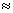 [H+]A
[H+]Aand from I equilibrium, [H+]A
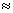 [HCO3-]
[HCO3-] 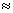 [H+]total
[H+]total[CO3-2] is negligible compared to [HCO3-] or [H+]total
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
A sample of hydrogen atoms in ground state is exposed to electromagnetic radiations of 1028 Å. The wavelengths of the in...Which of the following statements in relation to the hydrogen atom is correct?...The orbital diagram in which the aufbau principle is violated :...A positron is emitted from 23Na11 . The ratio of the atomic mass and atomic number of the resulting nuclide is...Consider an electron in the nth orbit of a hydrogen atom in the Bohr model. The circumference of the orbit can be expres...