SolutionHard
Question
If sodium sulphate is considered to be completely dissociated into cations and anions in aqueous solution, the change in freezing point of water (ᐃTf), when 0.01 mole of sodium sulphate is dissolved in 1 kg of water, is (Kf = 1.86 K kg mol-1)
Options
A.0.0372 K
B.0.0558 K
C.0.0744 K
D.0.0186 K
Solution
Vant Hoff′s factor (i) for Na2SO4 = 3
∴ ᐃTf = (i) kfm
= 3 × 1.80 ×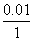 = 0.0558 K
= 0.0558 K
∴ ᐃTf = (i) kfm
= 3 × 1.80 ×
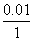 = 0.0558 K
= 0.0558 K Create a free account to view solution
View Solution FreeMore Solution Questions
The van′t Hoff factor i for an infinitely dilute solution of NaHSO4 is :...What is pH of the solution when 100 ml of 0.1M HCl is mixed with 100 ml of 0.1M CH3COOH....The phase diagrams for the pure solvent (solid lines) and the solution (non-volatile solute, dashed line) are recorded b...The osmotic pressures of 0.010 M solutions of KI and of sucrose (C12H22O11) are 0.432 atm and 0.24 atm respectively. The...Mole fraction of C3H5(OH)3 in a solution of 36 g of water and 46 g of glycerine is :...