Chemical Kinetics and Nuclear ChemistryHard
Question
The time for half life period of a certain reaction A → products is 1 hour. When the initial concentration of the reactant ′A′, is 2.0 mol L-1, how much time does it take for its concentration to come from 0.50 to 0.25 mol L-1 if it is a zero order reaction ?
Options
A.4 h
B.0.5 h
C.0.25 h
D.1 h
Solution
For a zero order reaction k = 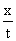 →(1)
→(1)
Where x = amount decomposed
k = zero order rate constant
for a zero order reaction
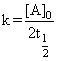 →(2)
→(2)
Since [A0] = 2M. t1/2 = 1 hr ; k = 1
∴ from equation (1)
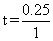 = 0.25 hr
= 0.25 hr
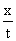 →(1)
→(1)Where x = amount decomposed
k = zero order rate constant
for a zero order reaction
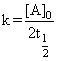 →(2)
→(2) Since [A0] = 2M. t1/2 = 1 hr ; k = 1
∴ from equation (1)
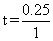 = 0.25 hr
= 0.25 hrCreate a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
Number of natural life times (Tav) required for a first-order reaction to achieve 99.9% level of completion is :...At 325°C, 1,3-butadiene dimerizes according to the equation: 2C4H6(g) → C8H12(g). It is found that the partial pressure ...If the rate of reaction, 2SO2(g) + O2(g) 2SO3(g) is given by :Rate = Kwhich statements are correct :...For a certain reaction involving a single reactant, it is found that $C_{0}\sqrt{T}$ is constant, where C0 is the initia...For the reaction system: 2NO(g) + O2(g) → 2NO2(g) volume is suddenly reduce to half its value by increasing the pr...