Nuclear Physics and RadioactivityHard
Question
The magnitude of angular momentum, orbit radius and frequency of revolution of electron in hydrogen atom corresponding to quantum number n are L, rand f resistively. Then according to Bohr′s theory of hydrogen atom
Options
A.fr2L is constant for all orbits
B.frL is constant for all orbits
C.f2rL is constant for all orbits
D.frL2 is constant for all orbits
Solution
L = 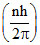 ; r =
; r = 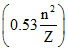 ; f ∝
; f ∝ 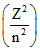
(f r L) ∝ Z Constant for all orbits.
(f r L) ∝ Z Constant for all orbits.
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
If radius of nucleus is estimated to be 3.6 Fermi then the radius nucleus be nearly...Half-life of a radioactive substance is 20 minutes. Difference between points of time when it is 33% disintegrated and 6...The half-life of $^{215}At$ is $100\ \mu s$. The time taken for the activity of a sample of $^{215}At$ to decay to $\dfr...10 gram of radioactive material of half-life 15 years is kept in a box for 20 years. The disintegrated material is :...When an elector-magnetic radiation is incident on the surface of metal, maximum .kinetic energy of photoelectric depends...