Nuclear Physics and RadioactivityHard
Question
The diagram shown the energy levels for an electron in a ace rain atom. Which transition shown represents the emission of a photon with the most energy ?
Options
A.III
B.IV
C.I
D.II
Solution
E4 - E3 = Z2 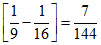 Z2
Z2
E2 - E3 = Z2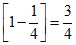 Z2
Z2
E4 - E2 = Z2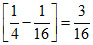 Z2
Z2
E2 - E3 = Z2
E4 - E2 = Z2
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Energy liberated in nuclear reaction is...A photo sensitive metallic surface has work function hv0. If photons of energy 2hv0 fall on this surface, the electrons ...Choose the CORRECT statement(s)...A radioactive nucleus (initial mass number A and atomic number Z) emits 3 α - particles and 2 positrons. The ratio ...The energy equivalent to 1 a.m.u. is :...