Nuclear Physics and RadioactivityHard
Question
A radioactive nucleus (initial mass number A and atomic number Z) emits 3 α - particles and 2 positrons. The ratio of number of neutrons to that of protons in the final nucleus will be
Options
A.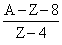
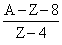
B.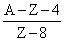
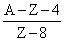
C.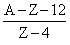
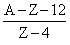
D.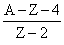
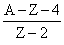
Solution
In positive beta decay a proton is transformed into a neutron and a positron is emitted.
p+ → n0 + e+
no. of neutrons initially was A - Z
no. of neutrons after decay (A - Z) - 3 × 2 (due to alpha particles) + 2 × 1 (due to positive beta decay)
The no. of proton will reduce by 8. [as 3 × 2 (due to alpha particles) + 2(due to positive beta decay)] Hence atomic number reduces by 8.
p+ → n0 + e+
no. of neutrons initially was A - Z
no. of neutrons after decay (A - Z) - 3 × 2 (due to alpha particles) + 2 × 1 (due to positive beta decay)
The no. of proton will reduce by 8. [as 3 × 2 (due to alpha particles) + 2(due to positive beta decay)] Hence atomic number reduces by 8.
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Which spectrum is continuous...An energy of 24.6 eV is required to remove one of the electrons forms a neutral from a neutral helium atom The energy (I...de-Broglie wavelength of an electron in the nth Boher orbit is λn and the angular momentum is Jn, then....Assume that the nuclear binding energy per nucleon (B/A) versus mass number (A) is as shown in the figure. Use this plot...The initial active nuclei of two radioactive substances are same and their half lives are 1 year and 2 year respectively...