Nuclear Physics and RadioactivityHard
Question
Choose the CORRECT statement(s)
Options
A. Mass of products formed is less that the original mass in nuclear fission and nuclear reactions.
B.Binding every per nucleon increases in α - decay and β - decay
C.mass number is conserved in all nuclear reactions.
D.atomic number is conserved in all nuclear reactions.
Solution
Fusion and fission are always exothermic and & decay will result in more stable product.
Mass number is conserved but atomic number is not conserved.
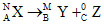 , = M + 0 and A may not be equal to B + C
, = M + 0 and A may not be equal to B + C
Mass number is conserved but atomic number is not conserved.
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Positron was discovered in the year :-...Which of the following nucleus is fissionable by slow neutrons :-...A $^{22}_{10}$Ne nucleus, after absorbing energy, decays into two $\alpha$-particles and an unknown nucleus. The unknown...The energy of a tungsten atom with a vacancy in L shell is 11.3 KeV. Wavelength of Kα photon for tungsten is 21.3 p...When four hydrogen nuclei combine to form a helium nucleus :-...