Nuclear Physics and RadioactivityHard
Question
Consider a hydrogen like atom whose energy in nth excited state is given by En = - 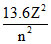 when this excited atom makes a transition from excited state to ground stat, most energetic photons have energy Emax = 52.2224 eV and least energetic photons have energy Emin = 1.224 eV. The atomic number of atom is -
when this excited atom makes a transition from excited state to ground stat, most energetic photons have energy Emax = 52.2224 eV and least energetic photons have energy Emin = 1.224 eV. The atomic number of atom is -
Options
A.2
B.5
C.4
D.None of these
Solution
Hence 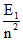 - E1 = 52.224eV and
- E1 = 52.224eV and 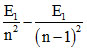 = 1.224eV ⇒ E1 = - 54.4eV and n = 5
= 1.224eV ⇒ E1 = - 54.4eV and n = 5
Now E1 = -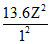 = - 54.4 eV. Hence Z - 2
= - 54.4 eV. Hence Z - 2
Now E1 = -
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
The half life of a radioactive element is 30days, in 90days the percentage of disintegrated part is...10 gram of radioactive material of half-life 15 years is kept in a box for 20 years. The disintegrated material is :...The magnitude of angular momentum, orbit radius and frequency of revolution of electron in hydrogen atom corresponding t...When a hydrogen atom is excited from ground state to first excited state, then...Initial ratio of active nuclei in two different samples is 2:3. Their half lives are 2hr and 3hr respectively. Ratio of ...