Nuclear Physics and RadioactivityHard
Question
When a hydrogen atom is excited from ground state to first excited state, then
Options
A.its kinetic energy increases by 10.2 eV
B.its kinetic energy increases by 13.6 eV
C.its potential energy increases by 10.2 eV
D.its angular momentum increases by h / 2π
Solution
In ground state, kinetic energy = 13.6 eV, potential energy = - 27.2eV
In first excited state, kinetic energy = 3.4 eV, Potential energy = - 6.8eV
Angular momentum is =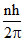 ; Difference of angular momentum for consecutive orbit =
; Difference of angular momentum for consecutive orbit = 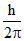
In first excited state, kinetic energy = 3.4 eV, Potential energy = - 6.8eV
Angular momentum is =
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Radius of which nucleus is half of radius of the nucleus Fe56 :-...The half life of a radioactive isotope X is 50 years. It decay to another element Y which is stable. The two elements X ...The maximum kinetic energy of photoelectrons emitted from a surface when photons of energy 6 eV fall on it is 4 eV. The ...The above is a plot of binding energy per nucleon Eb, against the nuclear mass M; A, B, C, D, E, F correspond to differe...When a hydrogen atom, initially at rest emits a photon resulting in transition n = 5 → n = 1. its recoil speed is ...