Nuclear Physics and RadioactivityHard
Question
The half life of a radioactive isotope X is 50 years. It decay to another element Y which is stable. The two elements X and Y were found to be in the ratio of 1 : 15 in a sample of a given rock. The age of the rock was estimated to be
Options
A.150 year
B.200 year
C.150 year
D.100 year
Solution
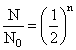
where n is number of half lives
∴
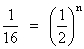 or
or 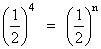 or n = 4
or n = 4Let the age of rock be t years
∵
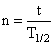
or, t = nT1/2 = 4 × 50 years = 200 years .
Create a free account to view solution
View Solution FreeMore Nuclear Physics and Radioactivity Questions
Two radiactive nuclie P and Q in a given sample decay into stable nucleus R. At time t = 0,number of P species are 4N0 a...The half life of a radioactive isotope is 2 hour, after which times it remains 1/64 times of its initial value...7/8 fraction of a sample disintegrates in t time. How much time it will take to disintegrete 15/16 fraction :-...The binding energy of deuteron is 2.2 MeV and that of 24He is 28 MeV. If two deuteron are fused to from one 24He than th...Mass number and atomic number of radioactive material are respectively 232 and 90. After disintegration mass number is 2...