SolutionHard
Question
For a dilute solution containing 2.5 g of a non-volatile non-electrolyte solute in 100 g of water, the elevation in boiling point at 1 atm pressure is 2oC. Assuming concentration of solute is much lower than theconcentration of solvent, the vapour pressure (mm of Hg) of the solution is (take Kb = 0.76 K kg mol-1)
Options
A.724
B.740
C.736
D.718
Solution
B → Solute; A → Solvent
WB = 2.5 g, WA = 100 g
ᐃTb = 2o
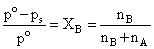
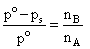 ∴ nB ≪ nA
∴ nB ≪ nA
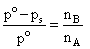
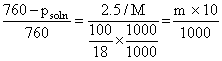 ....(i)
....(i)
and from boiling point elevation,
2 = 0.76 × m
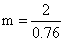 ....(ii)
....(ii)
on equating (i) and (ii)
Psoln = 724 mm
WB = 2.5 g, WA = 100 g
ᐃTb = 2o
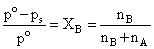
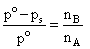 ∴ nB ≪ nA
∴ nB ≪ nA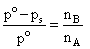
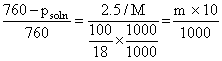 ....(i)
....(i)and from boiling point elevation,
2 = 0.76 × m
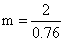 ....(ii)
....(ii)on equating (i) and (ii)
Psoln = 724 mm
Create a free account to view solution
View Solution FreeMore Solution Questions
A solution when diluted with H2O and boiled, it gives a white precipitate. On addition of excess NH4Cl/NH4OH. the volume...If P0 and P are the vapour pressures of a solvent and its solution respectively and N1 and N2 are the mole fractions of ...Which of the following 0.1 M aqueous solution will have the lowest freezing point ?...Liquids A and B form an ideal solution. The plot of $\frac{1}{X_{A}}$ (Y-axis) versus$\frac{1}{Y_{A}}$ (X-axis) (where X...The elevation in boiling point, when 13.44 g of freshly prepared CuCl2 is added to one kg of water, is (Some useful data...