Wave OpticsHard
Question
Based on equation E = -2.178 10-18 J 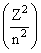 certain conclusions are written. Which of them is not correct ?
certain conclusions are written. Which of them is not correct ?
Options
A.For n = 1, the electron has a more negative energy than it does for n = 6 which means that the electron is more lossely bound in the smallest allowed orbit.
B.The negative sign in equation simply means that the energy of electron bound to the nucleus is lower than it would be if the electrons were at the infinite distance from the nucleus
C.Larger the value of n, the larger is the orbit radius
D.Equation can be used to calculate the change in energy when the electron change orbit
Solution
Correct answer is -
for n = 1 the electron has more negative energy than it does for n = 6 which means that the electron is less loosely bound in the smallest allowed orbit.
Create a free account to view solution
View Solution FreeMore Wave Optics Questions
Direction of the first secondary maximum in the Fraunhofer diffraction pattern at a single slit is given by (a is the wi...A monochromatic light source of wavelength λ is placed at S. Three slits S1, S2 and S3 are equidistant from the sou...The energy in the phenomenon of interference :...In Young′s double slit experiment, if monochromatic light is replaced by white light : -...light appears to travel in astright line, because...