Atomic StructureHard
Question
Energy required for the electron excitation in Li++ from the first to the third Bohr orbit is :
Options
A.36.3 eV
B.108.8 eV
C.122.4 eV
D.12.1 eV
Solution
En = - 13.6 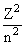
ELi++ = - 13.6 ×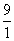 = - 122.4eV
= - 122.4eV
ELi++ = - 13.6 ×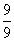 = - 13.6eV
= - 13.6eV
ᐃE = - 13.6 - (- 122.4)
= 108.8 eV
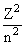
ELi++ = - 13.6 ×
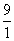 = - 122.4eV
= - 122.4eVELi++ = - 13.6 ×
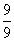 = - 13.6eV
= - 13.6eVᐃE = - 13.6 - (- 122.4)
= 108.8 eV
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In the Bohr model of a hydrogen atom, the centripetal force is furnished by the coulomb attraction between the proton an...Two thin conducting shells of radii R and 2R are shown in figure. The outer shell carries a charge +Q and inner shell is...When 5V potential difference is applied across a wire of length 0.1 m, the drift speed of electrons is 2.5 × 10-4 m...What is the energy of photon whose wavelength is 6840 ?...Light of wavelength 4000 is incident on a metal plate whose work function is 2eV.What is maximum kinetic energy of emitt...