Geometrical OpticsHard
Question
How many grams of concentrated nitric acid solution should be used to prepare 250 mL of 2.0 M HNO3?(70% HNO3 solution taken)
Options
A.
54.0 gm conc. HNO3
B.45.0 gm conc. HNO3
C.90.0 gm conc. HNO3
D.70.0 gm conc. NO3
Solution
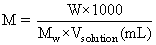
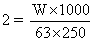
W = 31.5 g
70% HNO3 means 70g HNO3 is present in 100g solution.
∴ 31.5g HNO3 will be present in
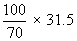
= 45 g of solution
Create a free account to view solution
View Solution FreeMore Geometrical Optics Questions
The plane faces of two identical plano-convex lenses, each having a focal length of 50 cm are placed against each other ...In the displacement method, a convex lens is placed in between an object and a screen. If one of the magnification is 3 ...Lenticels are involved in :...Yallow light is used in a sinle slit diffraction experiment with slit width of 0.6 mm. If yellow light is replaced by X-...Image of an object approaching a convex mirror of radius of curvature 20 m along its optical axis is observed to move fr...