Heat and Thermal ExpansionHard
Question
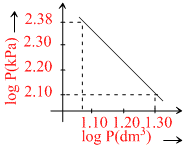
Logarithms of readings of pressure and volume fo ran ideal gas plotted on a graph as shown in figure. By measuring the gradient, It can be shown that the gas may be : -

Options
A.Monoatomic and undergoing an adiabatic change.
B.Monotomic and undergoing an isothermal change
C.Diatomic and undergoing an adiabatic change
D.Triatomic and undergoing an isothermal change
Solution
PVγ = C; ln P + γlnV = lnC
⇒ lnP = - γlnV = lnC ⇒ Y = mx + c
m = - γ = -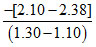 = - 1.4
= - 1.4
∴ The gas is diatomic
⇒ lnP = - γlnV = lnC ⇒ Y = mx + c
m = - γ = -
∴ The gas is diatomic
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
In the previous question, If the specific latent heat of vaporization of water at 0oC is time the specific latent heat o...A spherical body with an initial temperature T1 is allowed to coll in surrounding at temperature T0(< T1). The mass o...Two absolute absolute scales X and Y assigned numerical values 200 and 450 to the triple point of water.What is the rela...The temperature of a body rises by 44oC when a certain of heat is given t it. The same heat when supplied to 22 g of ice...A steel tape is placed around the earth at the equator when the temperature is 10oC. What will be the clearance between ...