Heat and Thermal ExpansionHard
Question
In the previous question, If the specific latent heat of vaporization of water at 0oC is time the specific latent heat of freezing of water at 0oC. the fraction of water that will ultimately freeze is.
Options
A.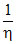
B.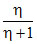
C.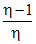
D.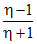
Solution
ᐃQvap = ᐃQfreezing
m (ηL) = M (L) ⇒ M = η
L = latent heat of freezing
m = mass of vapour
M = mass of freexed
∴ Fraction of water which freezed
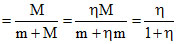
m (ηL) = M (L) ⇒ M = η
L = latent heat of freezing
m = mass of vapour
M = mass of freexed
∴ Fraction of water which freezed
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Two identical square rods of metal are end are welded end to end as shown in figure (a) 20 calories flows through it in ...The thermistors are usually made of...A gas undergoes an adiabatic process in which pressure becomes times and volume become of initial volume. If initial abs...Two identical beakers are filled with to the same level at 4oC. If one say A is heated while the other B is colled, then...Which of the following statements is /are correct ?...