Heat and Thermal ExpansionHard
Question
When unit of water boils to become steam at 100oC, it absorbs Q amount of heat. The densities o water and steam at 100o C are ρ1 and ρ2 respectively and the atmospheric pressure is ρ0. The increase in internal energy of the water is : -
Options
A.Q
B.Q + P0 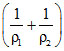
C.Q + P0 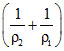
D.Q - P0 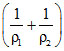
Solution
Q = ᐃU + W ⇒ +Q = ᐃU + P0 (V0 - V0)
⇒Q = ᐃU + P0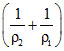 ᐃU = Q + P0
ᐃU = Q + P0 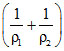
⇒Q = ᐃU + P0
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
A closed vessel contains a mixture of two diatomic gases A and B. molear mass of A is 16 time that of B and mass of gas ...A steel scale is to be prepared such that the millimeter intervals are to be accurate4 within 6 × 10-5 mm.The maxim...A thermocouple of negligible resistance produces an e.m.f of 40 μ V/oC in the linear range of temperature. A galvan...Which of the following statements is /are correct ?...A metal rod A of length by ᐃl when its temperature is raised by 100oC. Another rod B of different metal of length ...