Heat and Thermal ExpansionHard
Question
A closed vessel contains a mixture of two diatomic gases A and B. molear mass of A is 16 time that of B and mass of gas A contained in the vessel is 2 times of B. which of the following statements are true ?
Options
A.Average kinetic energy per molecule of A is equal to that of B
B.Root mean square value of translation velocity of B four times of A
C.Pressure exerted by B is eight time of that exerted A
D.Number of molecules of B in the in the cylinder is eight times that of A
Solution
Average KE per molecule in A& B = 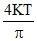
(vrms)A =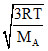 ; (vrms)B =
; (vrms)B = 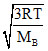
⇒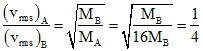
No. of mole of A =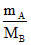
No. of mole of B =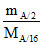 = 8nA
= 8nA
Pressure exerted by a gas in the vessel depends on the number of molecules present inside.
(vrms)A =
⇒
No. of mole of A =
No. of mole of B =
Pressure exerted by a gas in the vessel depends on the number of molecules present inside.
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Four moles of hydrogen , teo moles of helium and one mole of water vapor from an ideal gas mixture. What is the molar sp...Pressure versus temperature graph of an ideal gas is shown in figure. Density of the gas at point A is. ρ0 Density ...A block body emits at the rate P when its temperature is T. At this temperature the wavelength at which the radiation ha...Radiation from a black body at the thermodynamic temperature T1 is measured by a small detector at distance d1 from it. ...The figure shows two rods, one made of aluminum [α = 23 × 10-6 (Co)-1] and the other of steel [α = 12 ...