Atomic StructureHard
Question
A metal oxide has the formula Z2O3. It can be reduced by hydrogen to give free metal and water. 0.2 g of the metal oxide requires 12 mg of hydrogen for complete reduction. The atomic weight of the metal is
Options
A.52
B.104
C.26
D.78
Solution
Z2O3 + 3H2 → 2z + 3H2O
[0.2 g = 200 mg]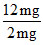
= 6 m mol
Since H2 used is 6 m mol, Z2O3 used should be 2 m mol.
Mol of Z2O3 =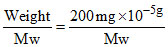 = 2 × 10-3 moles
= 2 × 10-3 moles
∴ Mw(Z2O3) = 100
∴ 2z + 16 × 3 = 100
z =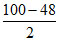 = 26
= 26
[0.2 g = 200 mg]
= 6 m mol
Since H2 used is 6 m mol, Z2O3 used should be 2 m mol.
Mol of Z2O3 =
∴ Mw(Z2O3) = 100
∴ 2z + 16 × 3 = 100
z =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Ground state electronic configuration of nitrogen atom can be represented by :...What is likely to be orbit number for a circular orbit of diameter 20 nm of the hydrogen atom if we assume Bohr orbit to...Photon having energy equivalent to the binding energy of 4th state of He+ ion is used to eject an electron from the meta...Which of the following pairs of ions are isoelectronic and isostructural ?...The fraction of volume occupied by the nucleus with respect to the total volume of an atom is...