Redox and Equivalent ConceptHard
Question
To 1 L of 1.0 M impure H2SO4 sample, 1.0 M NaOH solution was added and a plot was obtained as follows:
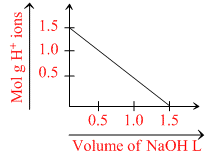

The % purity of H2SO4 and the slope of curve, respectively, are;
Options
A.75% - 1/2
B.75% - 1
C.50% - 1/3
D.50% - 1/2
Solution
i. Moles of NaOH = 1.0 M × 1.5 L = 1.5 mol
Moles of H2SO4 = 1.0 M × 1L = 1.0 mol
2 NaOH + H2SO4 → Na2SO4 + 2H2O
2mol 1 mol
Moles of H2SO4 reacted with NaOH
=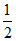 mol of NaOH
mol of NaOH
=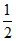 × 1.5 = 0.75 mol H2SO4
× 1.5 = 0.75 mol H2SO4
%purity of H2SO4 =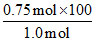 = 75%
= 75%
ii. For slope: = 1
Slope =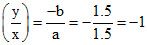
Moles of H2SO4 = 1.0 M × 1L = 1.0 mol
2 NaOH + H2SO4 → Na2SO4 + 2H2O
2mol 1 mol
Moles of H2SO4 reacted with NaOH
=
=
%purity of H2SO4 =
ii. For slope: = 1
Slope =
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
Which of the following properties are same for N2H2, N2F2 and H2N2O2....A small amount of CaCO3 completely neutralizes 525 ml of 0.1 N-HCl and no acid is left at the end. After converting all ...The oxisation number of carbon in CO2O is :...When copper is treated with a certain concentration of nitric acid, nitric oxide and nitrogen dioxide are liberated in e...Which of the following will be redox change :-...