Redox and Equivalent ConceptHard
Question
Which of the following properties are same for N2H2, N2F2 and H2N2O2.
Options
A.All are having two stereo isomer.
B.All are having equal number of bonds between two N-atoms.
C.All are having equal bond strength of N-N bond.
D.Oxidation state of N-atoms is same for all.
Solution
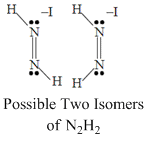
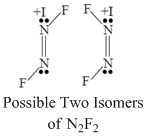
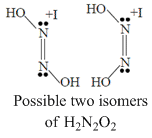
⇒ Due to having different substituents N-
N bond strength is not equal for all.
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
How many grams of H2S will react with 6.32 g KMnO4 to produce K2SO4 and MnO2? (K = 39, Mn = 55)...In the Haber’s process, the equivalent weight of ammonia is...Oxidation number of Cr in CrO5 is :-...A quantity of 1.878 g of a metal bromide when heated in a stream of hydrogen chloride gas is completely converted to the...Which of the following oxide can not act as reducing agent ?...