Heat and Thermal ExpansionHard
Question
A thermally insulated vessel contains some water at 0oC. The vessel connected to a vacuum pump out water vapor. This results in some water getting frozen . It is given latent heat if vaporization of water at 0oC = 21 × 105 J / kg and latent heat of freezing of water = 3.36 × 105 J / kg. The maximum percentage amount of water that will be solidified in this manner will be : -
Options
A.86.2%
B.33.6%
C.21 %
D.24.36%
Solution
Let x = percentage of water solidified then heat lost = heat gained
⇒ x × 3.36 × 105 = (100 - x) × 21 × 105
⇒ x =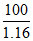 = 86.2%
= 86.2%
⇒ x × 3.36 × 105 = (100 - x) × 21 × 105
⇒ x =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
N(< 100) Molecules of a gas have velocities 1, 2, 3.........N , km/s respectively . Then :-...One mole of an ideal monatomic gas is taken from A to C along the path ABC. The temperature of the gas at A is T0. For t...If water at 0oC , kept in a container with an open top, is placed in a large evacuated chamber :-...N(< 100) Molecules of a gas have velocities 1, 2, 3.........N , km/s respectively . Then :-...The variation of lengths of two metal rods A and B with change in temperature is shown in figure. The ratio of is : -...