JEE AdvancedRedox and Equivalent ConceptHard
Question
1 g of the carbonate of a metal was dissolved in 25 mL of 1N-HCl. The resulting liquid required 5 mL of 1N-NaOH for naturalization. The Equivalent wt of the metal carbonate is
Options
A.50
B.30
C.20
D.None
Solution
Total HCl ⇒ 25 × 1 = 25 m Eq
Excess HCl ⇒ 5 × 1 = 5 m Eq
HCl used ⇒ 25 - 5 = 20 m Eq
∴ m Eq = of HCl = m Eq of carbonate
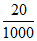 Eq =
Eq = 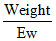 of carbonate = 1/E
of carbonate = 1/E
∴ E =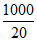 = 50
= 50
Excess HCl ⇒ 5 × 1 = 5 m Eq
HCl used ⇒ 25 - 5 = 20 m Eq
∴ m Eq = of HCl = m Eq of carbonate
∴ E =
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
A volume of 100 ml of 0.1 M-NaAl(OH)2CO3 is neutralized by 0.25 N-HCl to form NaCl, AlCl3 and CO2. The volume of HCl req...When K2Cr2O7 is converted into K2CrO4, the change in oxidation number of Cr is...In the standardization of Na2S2O3 using K2Cr2O7 by iodometry, the equivalent weight of K2Cr2O7is :...Standard reduction electrode potentials of three metals A,B&C are respectively +0.5 V, - 3.0 V & - 1.2 V. The reducing, ...The sum of oxidation states of all carbon atoms in toluene molecule is...