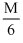Redox and Equivalent ConceptHard
Question
In the standardization of Na2S2O3 using K2Cr2O7 by iodometry, the equivalent weight of K2Cr2O7is :
Options
A.molecular weight /2
B.molecular weight /6
C.molecular weight /3
D.same as molecular weight
More Redox and Equivalent Concept Questions
Which of the following reaction is an example of comproportionation reaction?...The equivalent weight of H2SO4 in the reaction: 2KMnO4 + 3H2SO4 + 10HCl → 2MnSO4 + K2SO4 + 5Cl2 + 8H2O, is...In which of the following compound, the oxidation state of sulphur is +7?...A metal carbonate on heating is converted to metal oxide and is reduced to 60% of its original weight. The equivalent we...In the Haber’s process, the equivalent weight of ammonia is...