Atomic StructureHard
Question
Let υ1 be the frequency of the series limit of the Lyman series, υ2 be the frequency of the first line of the Lyman series, and υ3 be the frequency of the series limit of the Balmer series -
Options
A.υ1 - υ2 = υ3
B.υ2 - υ1 = υ3
C.υ3 = 1/2 (υ1 - υ3)
D.υ1 + υ2 = υ3
Solution
v = RC Z2 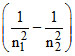 .
.
v1 = RC Z2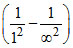 = RC Z2 , v2 = RC Z2
= RC Z2 , v2 = RC Z2 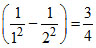 RC Z2.
RC Z2.
v3 = RC Z2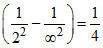 RC Z2. ∴ υ1 - υ2 = υ3.
RC Z2. ∴ υ1 - υ2 = υ3.
v1 = RC Z2
v3 = RC Z2
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
An electron and a proton are accelerated through a potential V. If Pe and Pp are their momentum, then PP: Pe ratio is ap...Which of the following ion have the same number of unpaired electrons as in Fe2+ (Z = 26)?...For hydrogen atom, the number of revolutions of the electron per second in the orbit of quantum number, n, is proportion...If wavelength is equal to the distance travelled by the electron in one second, then -...The average lifetime of an electron in an excited state of hydrogen atom is about 10–8 s. How many revolutions does an e...