Hydrogen and its compoundHard
Question
100 g sample of clay (containing 19% H2O, 40% silica. and inert impurities as rest) is partially dried so as to contain 10% H2O.
Which of the following is/are correct statements(s) ?
Which of the following is/are correct statements(s) ?
Options
A.The percentage of silica in it is 44.4 %.
B.The mass of partially dried clay is 90.0 g.
C.The percentage of inert impurity in it is 45.6%
D.The mass of water evaporated is 10.0 g.
Solution
Silica H2O Impurities
% in original clay] ⇒ 40 19 100 - (40 + 19) = 41
% after partial drying] ⇒ a 10 100 - (a + 10) = 90 - a
On heating, only water evaporates from clay, whereas silica and impurities are left as it is. Therefore, % ratio of silica and impurities remains unchanged i.e.,
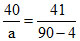 , ∴ a = 44.4 %
, ∴ a = 44.4 %
% of impurities after partial drying = (90 - a) = (90 - 44.4) = 45.6%
Mass of H2O evaporated = (19 - 10) = 9 g
% in original clay] ⇒ 40 19 100 - (40 + 19) = 41
% after partial drying] ⇒ a 10 100 - (a + 10) = 90 - a
On heating, only water evaporates from clay, whereas silica and impurities are left as it is. Therefore, % ratio of silica and impurities remains unchanged i.e.,
% of impurities after partial drying = (90 - a) = (90 - 44.4) = 45.6%
Mass of H2O evaporated = (19 - 10) = 9 g
Create a free account to view solution
View Solution Free