Hydrogen and its compoundHard
Question
How many unpaired electrons are present in Ni2+ ?
Options
A.0
B.2
C.4
D.8
Solution
Ni2+ = [Ar] 3d8, 4s0 and 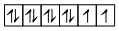
Thus t three are two unpaired electrons.
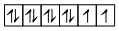
Thus t three are two unpaired electrons.
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
propane on hydroboration and oxidation producesγ...Amongst Ni(CO)4, [Ni(CN)4]2- and NiCl42- are :...″Metals are usually not found as nitrates in their ores″. Out of the following two (a and b) reasons which i...Thealkene R - CH - CH2 reacts readily with B2H6 and formed the product B which on oxidation with aikline hydrogen peroxi...With ammoncal cuprous chloride solution, a reddish precipitate is obtained on treatingwith:...