Hydrogen and its compoundHard
Question
(CH3)2C = CHCH3 + NOBr → product. The structure of the product is
Options
A.(CH3)2 C(NO) - CH(Br)CH3
B.(CH3)2 C(Br) - CH(NO)CH3
C.(CH3)2 CH - C(NO)(Br)CH3
D.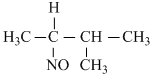
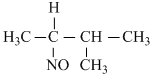
Solution
The reaction follows Markown ikoff rule namely the bromide ion adds on to the carbon having the least number of hydrogen and the more positive part namely the -NO group adds to the other double bond
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
In the following sequence of the reactions, what is D ?...Which one of the following processes will produce hard water?...Which of the following compounds are the common product/s obtained in the hydrolysis of XeF6 and XeF4 :...Calculate the amount of ammonium chloride required to dissolve in 500 ml water to have pH = 4.5 (Kb for NH4OH is 1.8 ...Which orbit of Be+3 has the same orbit radius as that of the ground state of hydrogen atom :-...