Atomic StructureHard
Question
For an electron, with n = 3 has only one radial node. The orbital angular momentum of the electron will be
Options
A.0
B.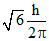
C.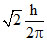
D.3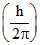
Solution
Number of radial nodes = n - l - 1 = 1, n = 3. ∴ l = 1
Orbital angular momentum =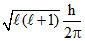 =
= 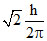
Orbital angular momentum =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In the structure of ClF3, the number of lone pairs of electrons on central atom 'Cl' is...The first use of quantum theory to explain the structure of atom was made by :...Deuterium nucleus contains :...The wavelength of a 150 g rubber ball moving with velocity of 50 ms-1 is :...Which of the following exists as covalentcrystals in the solid stae ?...