Redox and Equivalent ConceptVery Hard
Question
In alkaline medium, ClO-3 oxidises H2O2 to O2 and is itself reduced to Cl-. How many moles of H2O2 are oxidised by 1 mol of ClO-3 ?
Options
A.1
B.3/2
C.5/2
D.7/2
Solution
5e- + ClO-3 → Cl-
Eq of ClO-3 = Eq of H2O2
(n-factor = 5) (n-factor = 2)
1 mol of ClO-3 =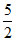 mol of H2O2
mol of H2O2
Eq of ClO-3 = Eq of H2O2
(n-factor = 5) (n-factor = 2)
1 mol of ClO-3 =
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
In the following reaction (unbalanced), equivalent weight of As2S3 is related to its molecular weight, M, byAs2S3 + H+ +...The pair of compounds having metals in their highest oxidation state is...In a reaction, calcium phosphate is producing calcium hydrogen phosphate. The equivalent weight of calcium phosphate in ...The specific heat of a metal is found to be 0.03. 10 g of the metal on treatment with nitric acid gave 18.9 g of pure dr...A quantity of 1 g of metal ion, M2+ was discharged by the passage of 1.81 × 1022 electrons. The atomic mass of the metal...