Redox and Equivalent ConceptHard
Question
A mole of N2H4 loses 10 mol of electrons to from a new compound y. Assuming that all the nitrogen appears in the new compound, what is the oxidation state of nitrogen in y ? (There is no change in the oxidation number of hydrogen.)
Options
A.-1
B.-3
C.+3
D.+5
Solution
N2H4 → 2y + 10e-
2x + 4 = 0 2y = 2x
2x = - 4
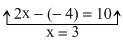
2x + 4 = 0 2y = 2x
2x = - 4
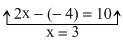
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
A volume of 50 ml of 0.1 M metal salt reacts completely with 25 ml of 0.1 M sodium sulphite. In the reaction, SO32− is o...For the redox reaction, Zn + NO3− → Zn2+ + NH4+ in basic medium, the coefficients of Zn, NO3− and OH− in the balanced eq...A quantity of 0.298 g of the chloride of a metal yielded 0.348 g of the sulphate of the same metal. The equivalent weigh...The oxidation state of C in C6H12O6 is equal to the oxidation state of C in...In the reaction, Cl2 + NaOH → NaCl + NaClO3 + H2O, the equivalent weight of H2O is...