Heat and Thermal ExpansionHard
Question
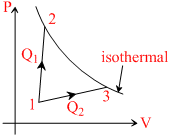
A gas takes part in two processes in which it is heated from the same initial state 1 to the same 1 to the same final temperature. The processes are shown on the P-V diagram by the straight line 1 - 2 and 1 - 3.2 and 3 are the points on the same isothermal curve. Q1 and Q2 are the heat transfer along the two processes. Then :-

Options
A. Q1 = Q2
B. Q1 < Q2
C. Q1 > Q2
D.insufficient data
Solution
Work done in process 1 - 3 is greater than that in process 1 - 2. while change in internal energy is same for both processes ∴ Q1 > Q2
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
When unit of water boils to become steam at 100oC, it absorbs Q amount of heat. The densities o water and steam at 100o ...Bars of two different metals are bolted together, as shown in figure. The distance x does not change with temperature if...n moles of an ideal triatomic linear gas undergoes a process in which the temperature changes with volume as T = k1V2 wh...An ideal monotonic gas undergoes a cycle ABCA as shown in the fig. The ratio of heat absorbed during AB to the work done...Figure shows the expansion of a 2m long metal rod temperature. The volume expansion coefficient meta is :-...