Heat and Thermal ExpansionHard
Question
n moles of an ideal triatomic linear gas undergoes a process in which the temperature changes with volume as T = k1V2 where k1 is a constant. Choose correct alternative (s).
Options
A.At normal temperature C = 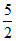 R
R
B.At normal temperature molar Cp - Cv = R
C.At normal temperature molar heat capacity C = 3R
D.At any temperature molar heat capacity C = 3R
Solution
At normal temperature Cv = 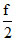 R =
R = 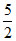 R ; At any temperature Cp - Cv =
R ; At any temperature Cp - Cv = 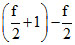 R = R from process T = k1V2 & ideal gas equation PV = nRT we have PV-1 = constant ⇒ x = - 1
R = R from process T = k1V2 & ideal gas equation PV = nRT we have PV-1 = constant ⇒ x = - 1
⇒ C = CV +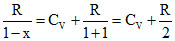 At normal temperature C =
At normal temperature C = 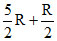 = 3R.
= 3R.
⇒ C = CV +
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Two conductors have the same resistance at 0oC but their temperature coefficients of resistance are α1 and α2....A inert gas obeys the law PVx = constant. For what value of x, it has negative molar specific heat -...540 g of ice at 0oC is mixed with 540g of water at 80oC. The temperature of the mixture is (Give latent heat of fusion o...A thermocouple of negligible resistance produces an e.m.f of 40 μ V/oC in the linear range of temperature. A galvan...A and Bare two points on a uniform metal ring whose center is C. The angle ABC = θ. A and B maintained at two diffe...