Heat and Thermal ExpansionHard
Question
When unit of water boils to become steam at 100oC, it absorbs Q amount of heat. The densities o water and steam at 100o C are ρ1 and ρ2 respectively and the atmospheric pressure is ρ0. The increase in internal energy of the water is : -
Options
A.Q
B.Q + P0 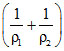
C.Q + P0 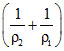
D.Q - P0 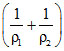
Solution
Q = ᐃU + W ⇒ +Q = ᐃU + P0 (V0 - V0)
⇒Q = ᐃU + P0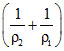 ᐃU = Q + P0
ᐃU = Q + P0 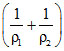
⇒Q = ᐃU + P0
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
Two wires are made of the same material and have the same volume. However wire 1 has crosssectional area A and wire-2 ha...A sonometer wire of length 1.5 m is made of steel. The tension in it produces an elastic strain of 1%. What is the funda...A mixture of n1 moles of monoatomic gas and n2 moles of diatomic gas has γ = 1.5 : -...Two plates of equal areas are in contact with each other. Their thickness are 2 cm ans 3cm respectively. Temperature of ...A gas is expanded volume V0 to 2V0 under three different process. Process 1 is isobaric process, process 2 is isothermal...