Heat and Thermal ExpansionHard
Question
One mole of an ideal monatomic gas is taken from A to C along the path ABC. The temperature of the gas at A is T0. For the process ABS : -
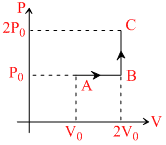

Options
A.Work done by the gas is RT0
B.Change in internal energy of the gas is 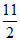 RT0.
RT0.
C.Heat absorbed by the gas is 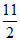 RT0.
RT0.
D.Heat absorbed by the gas is 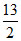 RT0.
RT0.
Solution
Work done = Area of ABC with V - axis
= P0(2V0 - 0) + 0 = P0V0 = nRT0 = RT0.
Change in internal energy = nCVᐃT
= 1 ×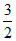 R × (4T0 - T0) =
R × (4T0 - T0) = 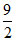 RT0
RT0
∴ Heat absorbed =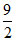 RT0 + RT0 =
RT0 + RT0 = 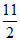 RT0.
RT0.
= P0(2V0 - 0) + 0 = P0V0 = nRT0 = RT0.
Change in internal energy = nCVᐃT
= 1 ×
∴ Heat absorbed =
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
In a process on a closed system of ideal gas, the initial pressure and volume is equal to the final pressure and volume...5 kg of steam at 100oC is mixed with 10 kg of ice at 0oC . Choose correct alternative /s(Given swater = 1 cal/goC, LF = ...Two substance A and B of equal mass m are heated by uniform rate of 6 cal s-1 similar conditions. A graph between temper...A steel scale measure the length of a copper rod as l0 when both are at 20oC, which is the calibration temperature for t...A gas takes part in two processes in which it is heated from the same initial state 1 to the same 1 to the same final te...