Heat and Thermal ExpansionHard
Question
5 kg of steam at 100oC is mixed with 10 kg of ice at 0oC . Choose correct alternative /s
(Given swater = 1 cal/goC, LF = 80 cal /g, LV = 540 cal/g)
(Given swater = 1 cal/goC, LF = 80 cal /g, LV = 540 cal/g)
Options
A.Equilibrium temperature of mixture is 160oC
B.Equilibrium temperature of mixture is 100oC
C.At equilibrium, mixture contains 13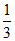 kg of water
kg of water
D.At equilibrium, mixture contains 13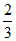 kg of water
kg of water
Solution
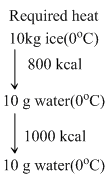

So available heat is more than required heat therefore final temperature will be 100oC.
Mass of heat condensed =
Total mass of steam = 5 -
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
The following are the P-V diagram for cyclic processes for a gas In which of these processes is heat absorbed by the gas...A gas is expanded volume V0 to 2V0 under three different process. Process 1 is isobaric process, process 2 is isothermal...Four containers are filled with monoatomic ideal gases. For each container, the number of moles, the mass of an individu...Four moles of hydrogen , teo moles of helium and one mole of water vapor from an ideal gas mixture. What is the molar sp...A wire elongates by l mm when a load W is hanged from it. If the wire goes over a pulley and two weights W each are hung...