Heat and Thermal ExpansionHard
Question
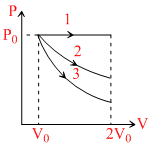
A gas is expanded volume V0 to 2V0 under three different process. Process 1 is isobaric process, process 2 is isothermal and 3 is adiabatic. let ᐃU1, ᐃU2 and ᐃU3 be the change internal energy of the is three process. Then:-

Options
A.ᐃU1 > ᐃU2 > ᐃU3
B.ᐃU1 < ᐃU2 < ᐃU3
C.ᐃU2 < ᐃU1 < ᐃU3
D.ᐃU2 < ᐃU3 < ᐃU1
Solution
ᐃU1 = +ve
ᐃU2 = 0
ᐃU3 = - ve
∴ ᐃU1 > ᐃU2 > ᐃU3
As volume increase, ᐃW = +ve
ᐃU2 = 0
ᐃU3 = - ve
∴ ᐃU1 > ᐃU2 > ᐃU3
As volume increase, ᐃW = +ve
Create a free account to view solution
View Solution FreeMore Heat and Thermal Expansion Questions
There is a small hole in a container. At temperature should it be maintained in order that it emits one calorie of energ...When water is heated from 0oC to 4oC and CP and CV are specific heats at constant pressure and constant volume respectiv...Two fine steel wires fastened between the projections of a heavy brass bar,are just taut when the whole system is at 0oC...Certain prefect gas is found to obey PVn = constant during adiabatic process. The volume expansion cofficient at tempera...Figure shows the expansion of a 2m long metal rod temperature. The volume expansion coefficient meta is :-...