Chemical EquilibriumHard
Question
For a reversible reaction aA + bB ⇋ cC + dD ; the variation of K with temperature is given by log 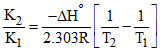 then
then
Options
A.K2 > K1 if T2 > T1 for an endothermic change
B.K2 < K1 if T2 > T1 for an endothermic change
C.K2 > K1 if T2 > T1 for an exothermic change
D.K2 < K1 if T2 > T1 for an exothermic change
Solution
For endothermic reaction, ᐃHo is positive so, if T2 > T1 then K2 > K1.
For exothermic reaction, ᐃHo is negative so, if T2 > T1 then K2 < K1.
For exothermic reaction, ᐃHo is negative so, if T2 > T1 then K2 < K1.
Create a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
Hydrolysis of sucrose gives :-Sucrose + H2O ⇋ Glucose + Fructose Equilibrium constant KC for the reaction is 2 ...Consider the following gaseous equilibrium in a closed container of volume "V" at T(K).$$P_{2}(\text{ }g) + Q_{2}(\text{...One mole of He (g, colourless) is added to the equilibrium mixture containing N2O4 (g, colourless) and NO2 (g, red-brown...What will the change on increasing the pressure at equilibrium water - water vapour?...A reaction at 300 K with ΔGo = −1743 J consists of 3 moles of A(g), 6 moles of B(g) and 3 moles of C(g). If A, B and C a...